Replication stress in stem cells and tissues
Background
Certain cell types and tissues are intrinsically more prone to replication stress, as they are associated with constitutive or inducible hyperproliferation. This requires cell division – and thus also full genome duplication – to occur repeatedly and accurately within a particularly short time. Besides its physiological relevance, hyperproliferation is also observed in precancerous lesions as a typical feature of tumorigenesis in many different tissues. Understanding the molecular mechanisms by which specific cell types adapt their replication process to their need to hyperproliferate - and how accelerated replication in early tumorigenesis contributes to genome instability and full transformation - is of crucial clinical relevance.
Goal
In this research line, we combine cell, molecular and structural biology methods to investigate the DNA replication process in specific cell types that constitutively hyper-proliferate or in tissues that are induced to hyper-proliferate in vivo under specific pathological conditions. The ultimate goal of our research is to uncover yet-elusive tissue-specific mechanisms of genome instability and tumorigenesis, linked to increased replication stress.
Ongoing and future work
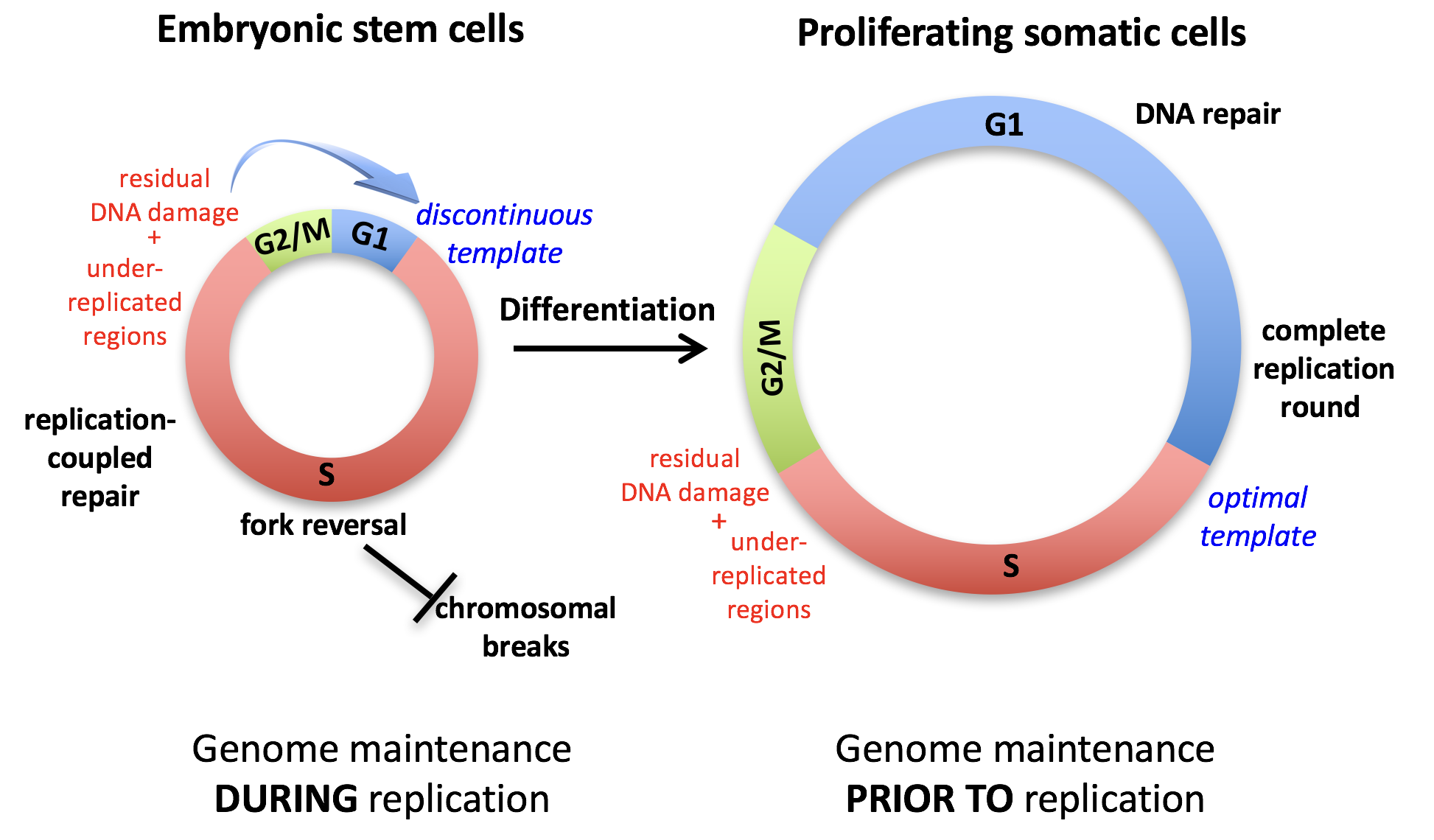
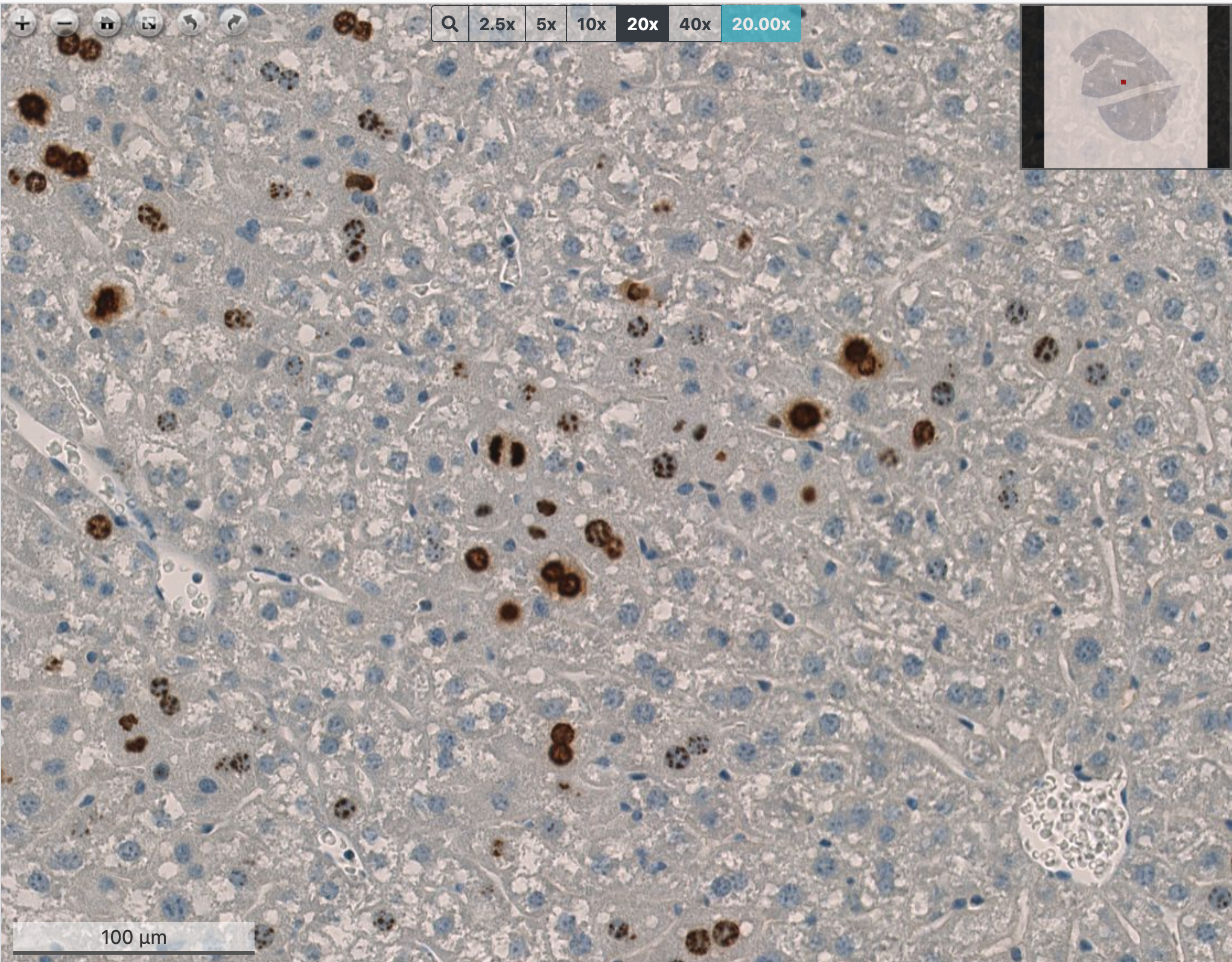
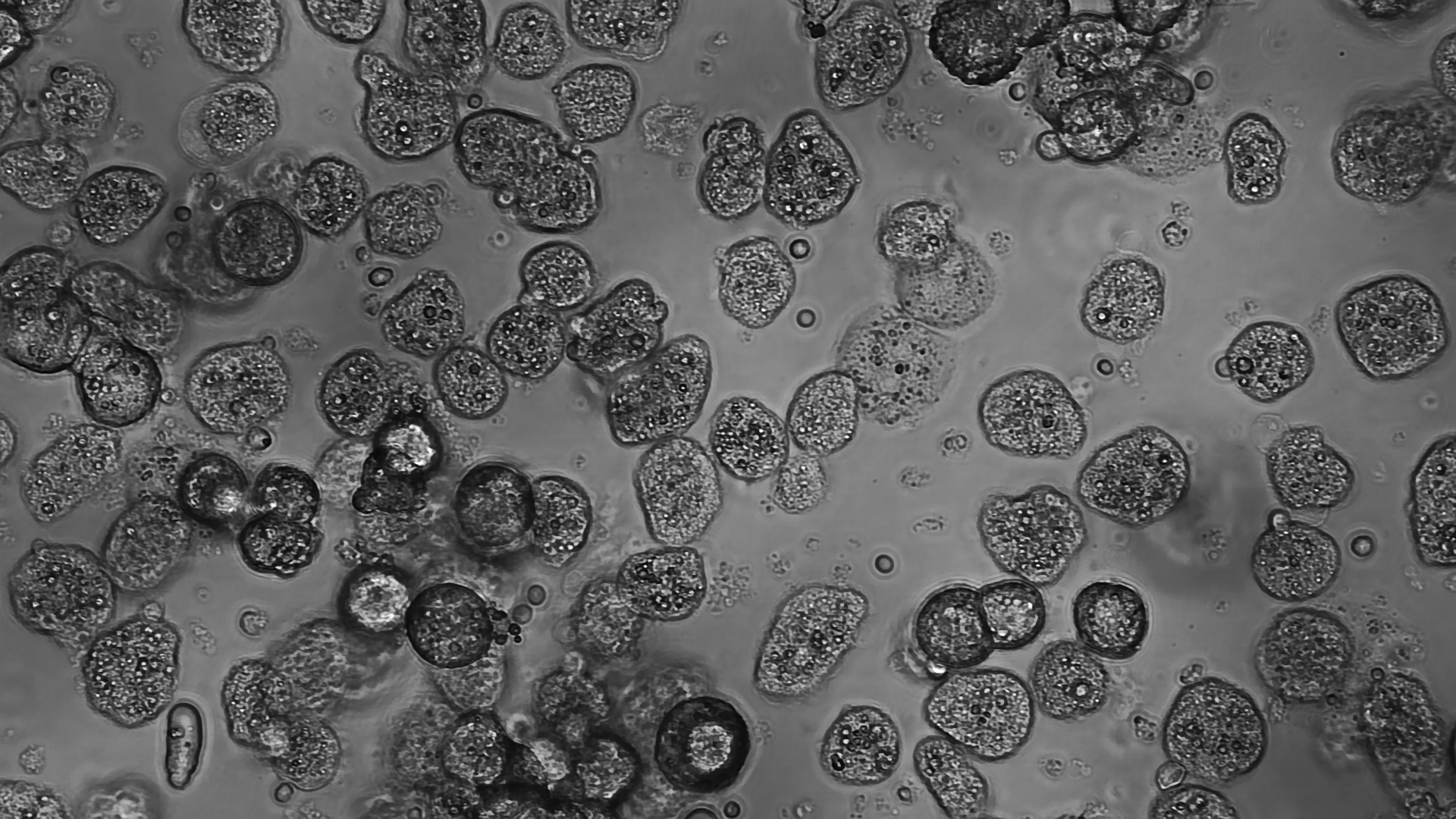
We reported how mouse embryonic stem cells – both in culture and in their natural environment – experience a peculiar form of replication stress, linked to the rapid succession of genome duplication rounds and short intervening gap phases (Ahuja et al., Nat Comms 2015). These fascinating studies have been recently extended to haematopoietic stem cells, which are usually quiescent, but need to undergo hyper-proliferative bursts in response to specific stimuli, displaying unexpected adaptations of the DNA replication process. In collaboration with clinical labs in Switzerland and abroad, we are now interested in elucidating molecular mechanisms contributing to hematological transformation and malignancies, that occur when stem and progenitor cells acquire specific signs of genomic instability and undergo uncontrolled proliferation. Similarly, in collaboration with Achim Weber’s group at our Institute, we are investigating the liver as prototype of conditionally hyper-proliferating tissue. This tissue – which is typically quiescent in healthy adults - can regenerate by acute hyper-proliferation upon partial hepatectomy and undergoes compensatory hyper-proliferation under cancer-associated chronic liver disease. Combining mouse models and patient-derived organoids – in collaboration with the University Hospital Zurich – we are currently exploring the impact of replication stress on liver tumorigenesis and its possible exploitation for therapeutic purposes. Overall, this research line has significantly expanded in recent years and represents at the moment one of our strongest areas of investigation in the lab.
Recent selected publications
K. Jacobs, C. Doerdelmann, J. Krietsch, D. González-Acosta, N. Mathis, S. Kushinsky, E. Guarino, C. Gomez-Escolar, D. Martinez, J.A. Schmid, P. J. Leary, R. Freire, A. R. Ramiro, C. M. Eischen, J. Mendez and M. Lopes (2022). Stress-triggered hematopoietic stem cell proliferation relies on PrimPol-mediated repriming. Molecular Cell, 82(21):4176-4188.e8. doi: 10.1016/j.molcel.2022.09.009
L.-A. Clerbaux, P. Cordier, N. Desboeufs, K. Unger, P. O. Leary, G. Semere, T. Y. Boege, L. K. Chan, C. Desdouets, M. Lopes and A. Weber (2023). Mcl-1 deficiency in murine livers leads to nuclear polyploidization and mitotic errors: implications for hepatocellular carcinoma. JHEP Rep. 2023 Jul 11;5(10):100838. doi: 10.1016/j.jhepr.2023.100838. eCollection 2023 Oct.